Sodium chloride is a highly soluble salt that has leached into the waters throughout the Earth’s history. The principal mineral that is mined for chlorine is halite (sodium chloride or ‘common salt’). Both of these compounds, however, are now closely regulated since they can harm the liver.Ĭhlorine is not found in nature uncombined. Chlorine was once widely utilised to manufacture anaesthetics like chloroform and carbon tetrachloride (a dry-cleaning solvent).At some point throughout the manufacturing process, chlorine or its derivatives are used in 85 percent of medications. It’s employed in substitution processes and as an oxidising agent. Organic chemistry is another important application for chlorine.Window frames, car interiors, electrical wire insulation, water pipes, blood bags, and vinyl flooring all employ this flexible plastic. PVC is made with about 20% of the chlorine produced.It’s also utilised in the production of hundreds of consumer goods, ranging from paper to paints, textiles to insecticides. It’s used to purify water for drinking and swimming pools. Chlorine is a disinfectant that kills bacteria.Unit Of Force- What Is The SI & CGS Unit Of Force?Ĭhlorine Atomic Mass and Weight in Kg and Gramsģ5Cl –Atomic Mass: 34.969 ( 75.76 %Natural abundance)ģ7Cl –Atomic Mass: 36.966 ( 24.24% Natural abundance).For Aadhar- New Registration, Update, Download.Class 12 Biology Term 2 Important Questions With Solutions.Area Of Parallelogram – Formula, Definition, Examples.Perimeter Of Triangle – Formula, Definition, Examples.As a result, the atomic mass of chlorine is determined to be 35.5u. The average mass of the naturally occurring isotopes of an element in comparison to the mass of an atom of 12C is known as relative atomic mass. The sum of the masses of protons, neutrons, and electrons in an atom or group of atoms is called atomic mass. 
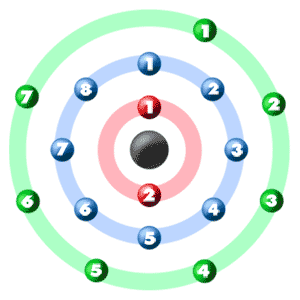
If there are 17 protons and 20 neutrons in a nucleus, about a quarter of the time, it happens in nature. If there are 17 protons and 18 neutrons in a nucleus, about 75% of the time, it happens in nature. A chlorine atom has 17 protons and is divided into two isotopes: chlorine 35 and chlorine 37. Isotopes are elemental varieties that have the same number of protons but differ in the number of neutrons in the atom. Chlorine Atomic Massīecause of the presence of isotopes, the atomic mass of chlorine is considered as 35.5u rather than 35u. The second-lightest of the halogens, it appears between fluorine and bromine in the periodic table and its properties are mostly intermediate between them. Chlorine is a chemical element with the symbol Cl and atomic number 17. Chlorine Atomic Mass and Weight in Kg and GramsĬhorine is a yellowy-green dense gas with a choking smell, which was discovered by Carl Wilhelm Scheele in 1774.Therefore, there are various non-equivalent definitions of atomic radius. 
However, this assumes the atom to exhibit a spherical shape, which is only obeyed for atoms in vacuum or free space. The atomic radius of a chemical element is a measure of the distance out to which the electron cloud extends from the nucleus. It must be noted, atoms lack a well-defined outer boundary. The atomic radius of Chlorine atom is 102pm (covalent radius). Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally-occuring isotopes and their abundance. 
The atomic mass is carried by the atomic nucleus, which occupies only about 10 -12 of the total volume of the atom or less, but it contains all the positive charge and at least 99.95% of the total mass of the atom. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Mass numbers of typical isotopes of Chlorine are 35 37. Isotopes are nuclides that have the same atomic number and are therefore the same element, but differ in the number of neutrons. The difference between the neutron number and the atomic number is known as the neutron excess: D = N – Z = A – 2Z.įor stable elements, there is usually a variety of stable isotopes. Neutron number plus atomic number equals atomic mass number: N+Z=A. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. Atomic Number – Protons, Electrons and Neutrons in ChlorineĬhlorine is a chemical element with atomic number 17 which means there are 17 protons in its nucleus. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
